

Products > TruDiagnosis® > TruArray MRSA Assay
The TruArray MRSA assay, in conjunction with components of the TruDiagnosis System, is a qualitative in vitro diagnostic assay for the direct detection of nasal colonization by Staphylococcus aureus (SA) and methicillin-resistant Staphylococcus aureus (MRSA) to aid in the prevention and control of MRSA infections. The assay utilizes an asymmetric polymerase chain reaction (PCR) for the simultaneous amplification and fluorescent labeling of SA/MRSA DNA that is recovered from a nasal swab specimen.
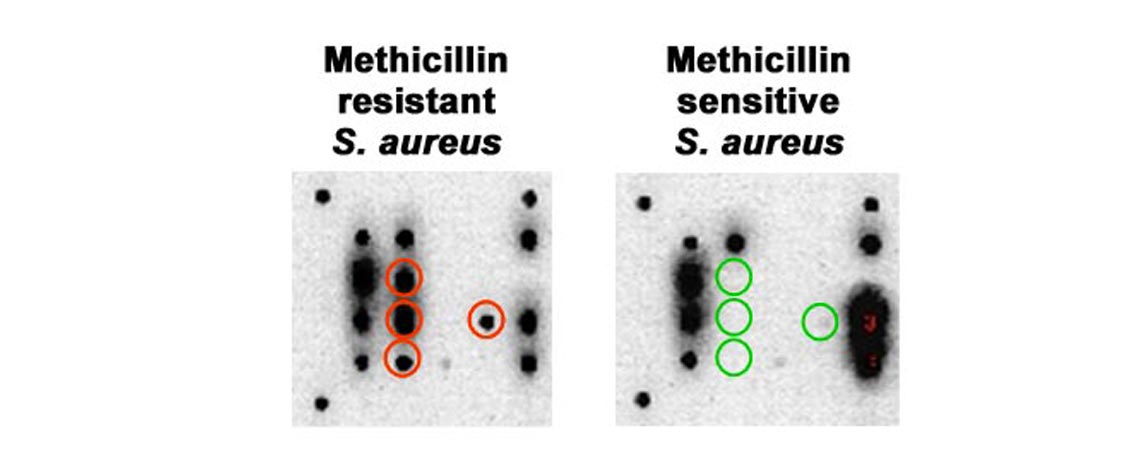
Sensitive and specific differentiation between MRSA and MSSA.
Zimmerman CE, Stamper PD, Bryant L, Farley J, Golova J, Holmberg R, Howard T, Linger Y, Meyers K, Perov A, Rudy GB, Carroll KC, Chandler DP. 2012. Development of a simple, low-density array to detect methicillin-resistant Staphylococcus aureus and mecA dropouts in nasal swabs. J Microbiol Methods. 91(3):366-76.
Cooney CG, Sipes D, Thakore N, Holmberg R, Belgrader P. 2012. A plastic, disposable microfluidic flow cell for coupled on-chip PCR and microarray detection of infectious agents. Biomed. Microdevices 14: 45-53.
* All of Akonni Biosystems tests are currently for Research Use Only, and are not for use in diagnostic procedures. Akonni is interacting with the Food & Drug Administration (FDA) in order to develop and prepare pre-market notifications seeking clearance to market these products as diagnostic assays in the U.S.
© 2003-2025 Akonni Biosystems, Inc. All rights reserved. Terms and Conditions • Privacy Policy
TruDiagnosis Systems, TruDx Readers, TruArray Tests, TruTip Kits, Primers, Lysis and Binding Buffers, Wash Buffers and Elution Buffers: For Research Use Only. Not for use in diagnostic procedures. No claim or representation is intended to provide information for the diagnosis, prevention or treatment of a disease.